Authors: Stephanie R. Ovalles , Jørn H. Hansen , and Huw M. L. Davies
Org. Lett.,
2011, 13 (16), 4284–4287
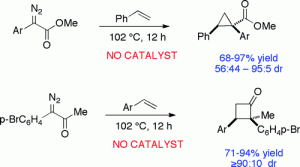
The thermal decomposition of aryldiazoacetates and aryldiazoketones in the absence of a catalyst leads to synthetically useful transformations. The thermal reaction of aryldiazoacetates with alkenes generates cyclopropanes in 68–97% yield and with good diastereoselectivity (up to 19:1 dr) when the aryl substituent is electron-rich. The thermal reaction of aryldiazoketones with alkenes generated cyclobutanones in 71–94% yield and with good diastereocontrol (≥9:1 dr).