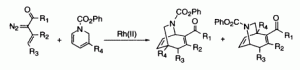Authors: Huw M.L. Davies, L. Mark Hodges, Craig T. Thornley
Tetrahedron Lett.,
1998, 39, 18, 2707-2710
A series of functionalized 6-azabicyclo[3.2.2]nonadienes was synthesized via rhodium(II)-catalyzed decomposition of vinyldiazomethanes in the presence of substituted dihydropyridines. The regioselectivity of the reaction was shown to be highly dependent on the steric bulk of the catalyst as well as the structure of the vinyldiazomethane. Each regioisomer was elaborated into higher homologs of ferruginine (1, 2).