MURI: Molecular Level Studies of Solid-Liquid Interfaces in Electrochemical Processes
The efficient interconversion of electrical and chemical energy requires functional electrodes that are able to catalyze complex multi-electron energy conversion reactions. The electrodes in energy conversion devices must, therefore, serve not merely as inert sources or sinks of electrons but rather as active players that bind 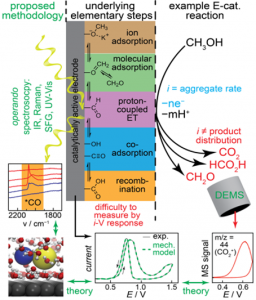 reactants, stabilize key intermediates, and enable inner-sphere electron transfer (ISET) reactions. Despite its fundamental and technological importance, a molecular-level mechanistic understanding of the factors that drive efficient multi-electron inner-sphere electrocatalysis remains elusive, impeding systematic progress toward innovative and more efficient energy conversion technologies. This critical knowledge gap arises because the current-voltage response of an electrode provides little information about the complex array of elementary surface processes that underpin near all energy conversion reactions. Addressing this longstanding grand challenge requires multi-faceted methodologies that go beyond classical current-voltage measurements and combine operando probes and atomistic simulations to develop multiscale models of critical ISET reactions.
reactants, stabilize key intermediates, and enable inner-sphere electron transfer (ISET) reactions. Despite its fundamental and technological importance, a molecular-level mechanistic understanding of the factors that drive efficient multi-electron inner-sphere electrocatalysis remains elusive, impeding systematic progress toward innovative and more efficient energy conversion technologies. This critical knowledge gap arises because the current-voltage response of an electrode provides little information about the complex array of elementary surface processes that underpin near all energy conversion reactions. Addressing this longstanding grand challenge requires multi-faceted methodologies that go beyond classical current-voltage measurements and combine operando probes and atomistic simulations to develop multiscale models of critical ISET reactions.

