Authors: Huw M. L. Davies and Brian D. Doan
J. Org. Chem.,
1999, 64 (23), 8501–8508
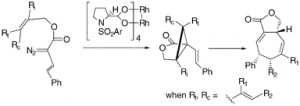
The asymmetric induction in the intramolecular cyclopropanations of allyl vinyldiazoacetates catalyzed by tetrakis[N-[4-dodecylphenyl)sulfonyl]-(S)-prolinato]dirhodium [Rh2(S-DOSP)4] is very dependent on the allyl substitution pattern. The reactions of cis-alkenes result in much higher asymmetric induction than trans-alkenes while the highest enantioselectivity was obtained with a disubstituted terminal alkene. The intramolecular cyclopropanation of dienylmethyl vinyldiazoacetates results in the synthesis of fused cycloheptadiene ring systems with full control of relative stereochemistry and variable enantioselectivity. The synthetic utility of this process was demonstrated by a short synthesis of 5-epi-tremulenolide in 93% ee.