Authors: James R. Manning, Tammy Sexton, Steven R. Childers and Huw M.L. Davies
Biorg. Med. Chem. Lett.,
2008, 19, 1, 58-61
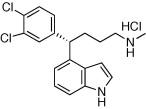
A series of enantiomerically pure 1-naphthyl and 4-indolyl arylalkylamines were prepared and evaluated for their binding affinities to the monoamine transporters. The two series of enantiomers displayed considerable differences in binding selectivity between the monoamine transporters, leading to the design of (S)-4-(3,4-dichlorophenyl)-4-(1H-indol-4-yl)-N-methylbutan-1-amine as a potent inhibitor for the dopamine and serotonin transporters.